The Process of Combustion
The combustion process is a fundamental chemical reaction that powers our world. At its core, combustion is a high-temperature exothermic reaction between a fuel and an oxidant, usually atmospheric oxygen. This reaction transforms chemical energy into thermal energy, liberating significant heat and light.
An exothermic reaction is a chemical process that releases energy into the surroundings in the form of heat or light, leading to an increase in the temperature and a negative enthalpy change.
Chemistry of Combustion
The basic chemical equation for combustion can be summarised as:
Fuel + O2 → Products + Heat
During this process, the internal chemical energy stored in the molecular bonds of the fuel is released. This release is measured as the heat of reaction i.e the quantity of heat exchanged between the system and its surroundings at a constant temperature.
Chemical Reactions and Energy Output
The efficiency and energy yield of combustion depend on the element being oxidised. Below are the standard energy releases for common fuel components:
| Element | Reaction | Heat Released (kcal/kg) |
|---|---|---|
| Carbon (Complete) | C + O2 → CO2 | 8,084 |
| Carbon (Incomplete) | 2C + O2 → 2CO | 2,430 |
| Sulphur | S + O2 → SO2 | 2,224 |
| Hydrogen | 2H2 + O2 → 2H2O | 28,922 |
The Role of Nitrogen in Combustion
While oxygen is the essential for combustion, it only makes up about 21% of our air. The remaining 79% is primarily nitrogen, which acts as a temperature reducing diluter.
Efficiency Loss: Nitrogen absorbs heat during the reaction, which reduces the overall combustion efficiency and the amount of usable heat.
Environmental Impact: At extreme temperatures, nitrogen reacts with oxygen to form nitrogen oxides (NOx). These are toxic pollutants and a primary focus of modern emission control technologies.
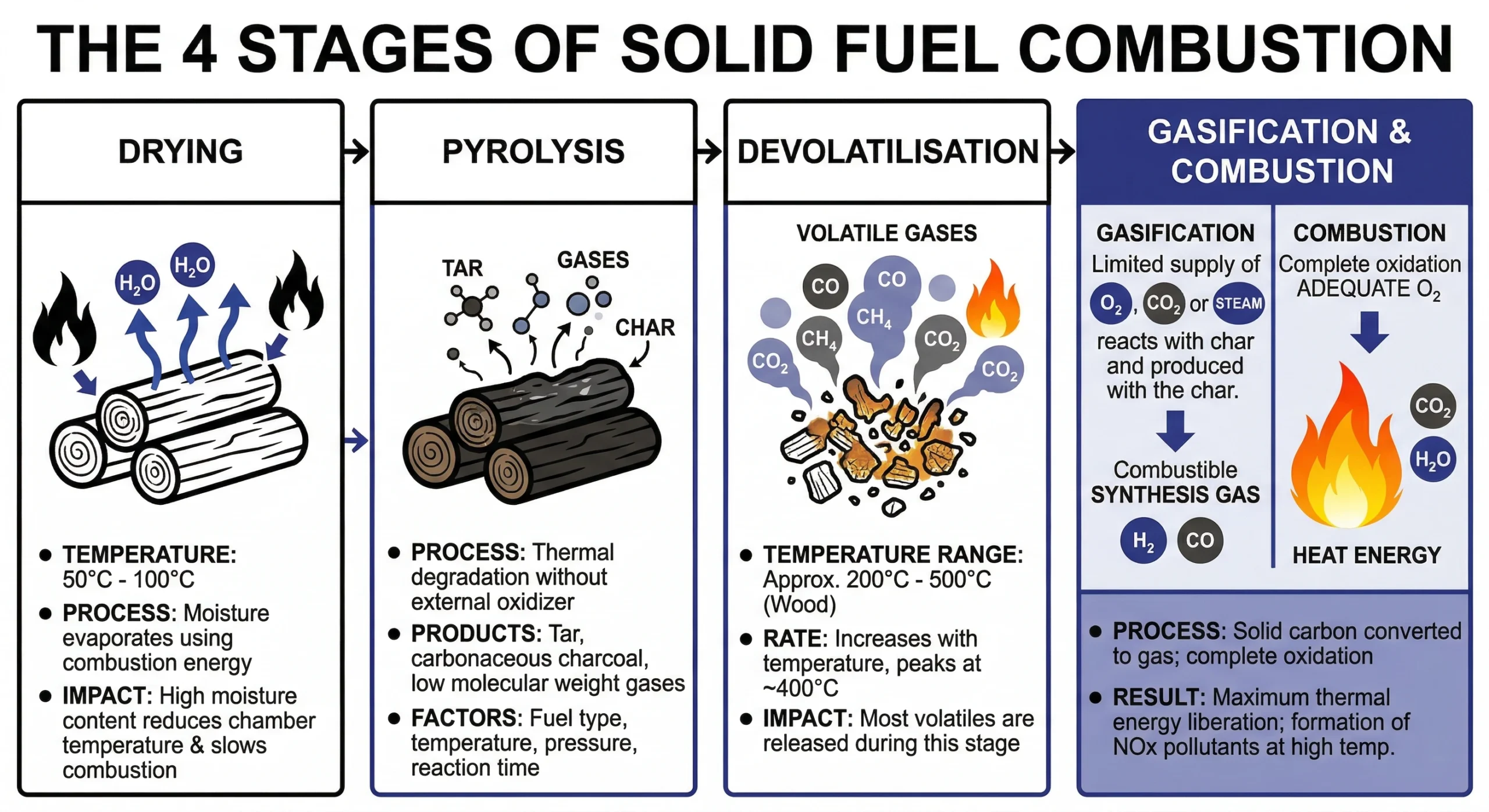
The 4 Stages of Solid Fuel Combustion
Combustion is not instantaneous, it happens in distinct, overlapping phases, especially in solid fuels like wood or coal.
1. Drying (Moisture Removal)
Before a fuel can burn, its moisture must evaporate. This typically occurs between 50°C and 100°C.
Note: High moisture content is known as a heat sink. It uses the energy released by the fire to turn water into steam, lowering the furnace temperature and slowing the entire process.
2. Pyrolysis
Pyrolysis is the thermal degradation of fuel in the absence of oxygen. This stage breaks down complex organic compounds into:
- Tars and carbonaceous charcoal (char).
- Low molecular weight gases.
- Small amounts of CO and CO2
3. Devolatilisation
In wood, devolatilisation begins around 200°C. The rate peaks at 400°C, where the majority of volatile gases are released. By 500°C, the rate slows significantly as the fuel transitions into its final char state.
4. Gasification
Unlike pyrolysis, gasification occurs in the presence of an oxidising agent (air, steam, or CO2). This stage converts the remaining carbonaceous solids into a gaseous fuel, ensuring that as much energy as possible is extracted from the source material.
Ideally, combustion ends in the complete oxidation of the fuel. However, factors like fuel type, pressure, and air-to-fuel ratios determine whether you achieve clean combustion or incomplete combustion which results in soot and dangerous carbon monoxide.
Infographic: The 4 stages of solid fuel combustion

